|
Of course it can interact with positively polarized hydrogens in an electrostatic way. So, it seems unlikely that the ammonium ion would be able to form any additional covalent bonds. 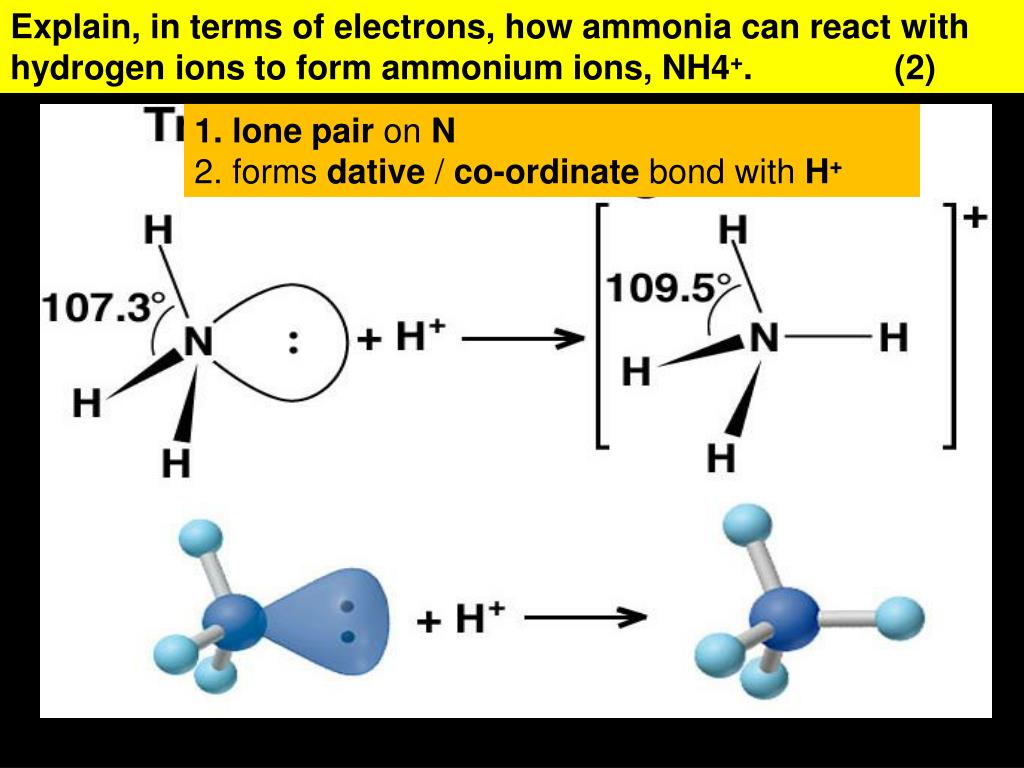
However, we also know that hydrogen bonding is partially covalent. It must be stored under pressure or at a low temperature. NH3 boils at 33.34 C (28.012 F) at standard pressure. It is also the simplest binary pnictogen hydride. Place the remaining two electrons on the nitrogen atom. Ammonium ion still has nitrogen bearing a partial negative charge. Ammonium ion still has nitrogen bearing a partial negative charge. Ammonia is referred to as a binary compound, meaning that it was formed from two non-equivalent types of atoms. Now draw a third hydrogen atom next to the nitrogen atom and place a third pair of electrons between these atoms. Now draw a second hydrogen atom next to the nitrogen atom and place a second pair of electrons between these atoms. The hydrogen's electron is left behind on the chlorine to form a negative chloride ion. 
The total number of electrons for ammonia will therefore be 8 electrons.ĭraw a hydrogen atom next to a nitrogen atom and place a pair of electrons between these two atoms. When the ammonium ion, NH4+, is formed, the fourth hydrogen is attached by a Coordinate covalent bond - Wikipedia, because only the hydrogen's nucleus is transferred from the chlorine to the nitrogen. 
The nitrogen atom has 5 electrons and each of the three hydrogen atoms has 1 electron. For example, the mutual attraction between an aspartic acid carboxylate ion and a lysine ammonium ion helps to maintain a particular folded area of a protein (part (a) of Figure 22.4. What are the numbers of bonding electrons for the nitrogen atom and the hydrogen atoms? Ionic bonds result from electrostatic attractions between positively and negatively charged side chains of amino acids. The arabic numeral above the element's column in the periodic table gives you that number. The first step in drawing the electron dot formula for ammonia is to determine the number of bonding electrons for each of the atoms. What is the first step that you must do in order to draw the electron dot formula for ammonia, NH 3? If you get stuck, try asking another group for help. There are therefore 4 pairs, all of which are bonding because of the four hydrogens. You should try to answer the questions without referring to your textbook. In this case, nitrogen contributes both bonding electrons, since the hydrogen ion has no electrons to begin with.\) The bond is called coordinate (or dative covalent) because both bonding electrons come from one of the atoms that form the bond. These two electrons are now bonding electrons between nitrogen and the fourth hydrogen atom in #"NH"_4^(+)#. So, there will be a total of 7 + 4 11 protons. Nitrogen has an atomic number of 7, and hydrogen has an atomic number of 1, and so the total atomic number of the hydrogens will be 1 4 4. The nitrogen atom donates its lone pair of electrons to form a coordinate bond with the proton. Explanation: In an ammonium ion (N H + 4) ion, there is one nitrogen atom and four hydrogen atoms. Using other terminology, ammonia acts as a nucleophile, which is a reactant that provides a lone pair of electrons for the formation of a covalent bond. What you're essentially dealing with here is the reaction between a Lewis base, which an electron donor, and a Lewis acid, which acts as an electron acceptor. The bond that is formed when a hydrogen ion is attached to the ammonia molecule is called a coordinate bond (or a dative covalent bond). Now, a hydrogen ion, #H^(+)#, which essentially is a proton, will be attracted to that lone pairs of electrons. Notice that you have a lone pair of electrons present on the nitrogen atom. The nitogen atom will bond with the hydrogen atoms via single bonds. The molecule has a total of 8 valence electrons, 5 from nitrogen and 1 from each of the three hydrogen atoms. To get a better understanding of how the ammonium ion, #"NH"_4^(+)#, is formed, start with the Lewis structure of ammonia.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
